|
12/31/2023 0 Comments Igor pro 8 no data was found in file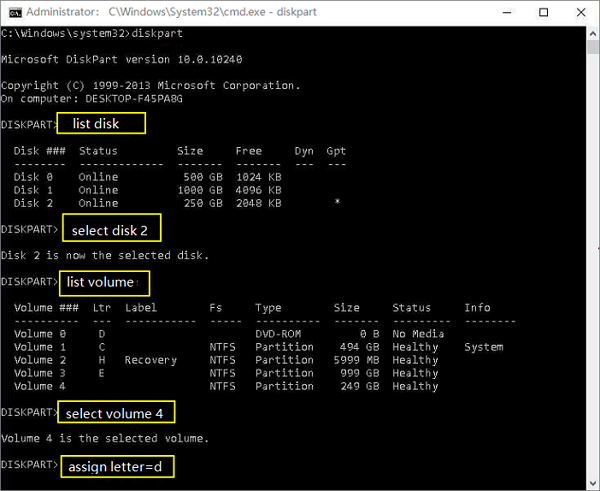
To understand the etiology of ASD symptoms, zebrafish is a powerful model system. 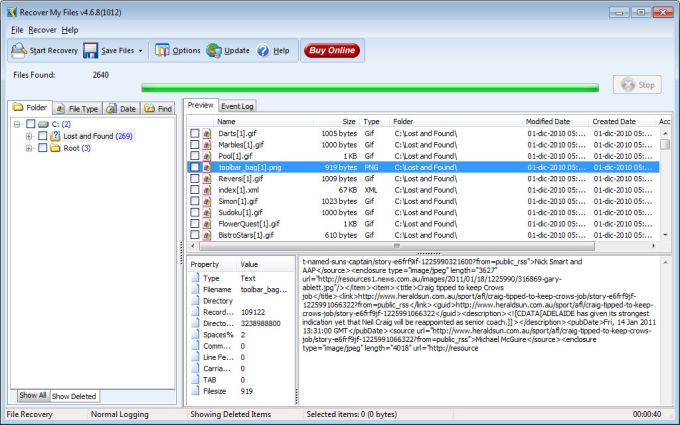
Studies to explore roles for SHANK3 in relation to GI dysfunction, however, are limited. Studies suggest that SHANK3 may also play important GI-related roles in host/symbiont interactions and Zn metabolism and intestinal barrier function. In addition, SHANK3 is also expressed at early developmental stages prior to synapse formation, as well as in enterocytes and nitrergic neurons of the enteric nervous system (ENS), and has been shown to have important interactions with the Wnt signaling pathway. SHANK3 is known to act as a synaptic scaffolding protein in the central nervous system (CNS) where it helps to regulate synaptic development, glutamatergic receptor signaling, actin polymerization, and dendritic spine formation. The majority of SHANK3 loss-of-function animal models are mammalian and have provided great insight into neural mechanisms related to social and motor behaviors characteristic of ASD. To investigate the biological mechanisms underlying GI distress in PMS and ASD, we have generated a zebrafish shank3 mutant model. In individuals with PMS, GI distress is characterized by reflux, cyclical vomiting, diarrhea, and/or constipation. 
Our work focuses on a monogenic form of ASD, Phelan-McDermid syndrome, that is caused by mutations that disrupt one copy of the SHANK3 gene resulting in SHANK3 haploinsufficiency. Consistent with prospective findings, clinical reports from monogenic causes of ASD regularly document GI distress. Despite the negative impacts of GI distress on daily life, ASD-associated GI symptoms are poorly understood. Here, we focus on gastrointestinal (GI) distress, one of the more frequent comorbidities experienced by individuals with ASD. While ASD diagnoses are based upon deficits in social communication and the presence of repetitive behaviors and/or restrictive interests, co-occurring symptoms (comorbidities) are also common. This is the first study to date demonstrating DT dysmotility in a zebrafish single gene mutant model of ASD.Īutism spectrum disorder (ASD) is estimated to impact more than 1% of the population and is etiologically and clinically heterogeneous. Reductions in serotonin-positive EECs and serotonin-filled ENS boutons suggest an endocrine/neural component to this dysmotility. Our data and rescue experiments support mutations in SHANK3 as causal for GI transit and motility abnormalities. Serotonin-positive enteroendocrine cells (EECs) were significantly reduced in both shank3abΔC +/− and shank3abΔC −/− mutants ( p < 0.05) while enteric neuron counts and overall structure of the DT epithelium, including goblet cell number, were unaffected in shank3abΔC +/− larvae. 
Rescue injections of mRNA encoding the longest human SHANK3 isoform into shank3abΔC +/− mutants produced larvae with intestinal bulb emptying similar to wild type (WT), but still deficits in posterior intestinal motility. Significantly slower rates of DT peristaltic contractions ( p < 0.001) with correspondingly prolonged passage time ( p < 0.004) occurred in shank3abΔC +/− mutants. Human SHANK3 mRNA was then used to rescue DT phenotypes in larval zebrafish. Because PMS is caused by SHANK3 haploinsufficiency, we assessed the digestive tract (DT) structure and function in zebrafish shank3abΔC +/− heterozygotes. To generate a zebrafish model of PMS, we used CRISPR/Cas9 to introduce clinically related C-terminal frameshift mutations in shank3a and shank3b zebrafish paralogues ( shank3abΔC). Here, we investigate the physiological basis for GI distress in ASD by studying gut function in a zebrafish model of Phelan-McDermid syndrome (PMS), a condition caused by mutations in the SHANK3 gene. For people with ASD, gastrointestinal (GI) distress is a commonly reported but a poorly understood co-occurring symptom. Figure 2 illustrates the data loading feature.Autism spectrum disorder (ASD) is currently estimated to affect more than 1% of the world population. Figure 4.8 illustrates some of these features. The file location can be accessed by the Open Folder button. Use the Control or Shift button together with a mouse click to select multiple traces. Clicking on the button (note the dropdown button ) saves only those traces that were selected by a mouse click. All measurements in the history list can be saved with. Double-clicking on a list entry allows you to rename it. The button to the left of each list entry controls the visibility of the corresponding trace in the plot the button to the right controls the color of the trace. By default, the plot area in those tools displays the last 100 measurements (i.e., depending on the tool, sweep traces, scope shots, DAQ data sets, or spectra), and each measurement is represented in as a list entry in the History sub-tab. Support feature saving, autosaving, and loading functionality. Such as Sweeper Tab, Data Acquisition Tab
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
